Moderna Case Study
Since its founding and unprecedented growth out of the pandemic, Moderna has established itself as a leading mRNA technology company–fueled by the requisite digital infrastructure and agile teams required to reimagine how medicines are created and delivered. The organization recently made headlines for partnering with OpenAI to advance mRNA medicine, and in general has a culture and practice of leading with a digital-first mindset in order to make the greatest impact for patients. Moderna’s internal mission, values, and mindsets drive its scientific progress and global workplace culture to build an organization that cares deeply for patients, employees, communities, and the environment.
Medical Affairs has an educational and data generation role as the bridge between Research and Commercial functions. With healthcare professionals as their typical primary stakeholders, they are experts at tailoring communications to the needs of medical doctors, policymakers, and others to make informed decisions and recommendations that influence individual and public health. Before the pandemic, teams aligned their narratives to a scientific communications platform (SCP) that was set at the beginning of the year and remained largely unchanged, but during the pandemic, the sheer volume of daily data releases and updates on the virus, its evolution, evidence on the most vulnerable populations, efficacy and durability, and overall real world evidence (RWE) was shooting out of a firehose, and the traditional “set it and forget” method for building an SCP needed to be reimagined. The Medical Affairs team needed to find a way to efficiently update their SCP to keep the research and data fresh.
The global Moderna Scientific Communications and Digital teams set out to find a way to incorporate new information into the SCP while maintaining a high level of certainty and trust in the information. They essentially needed a close to real-time solution for storing, sorting, and accessing libraries of information.
Solution requirements
Before researching solutions, the Moderna Medical Affairs team identified key questions and solution requirements to help employees quickly and easily find the research and data they needed.
The procurement team wanted to know:
- How can they create an agile SCP using a software-as-a-service platform that easily organizes new information and keeps research up-to-date?
- How can a new solution ensure research citations are always correct, especially since Medical Affairs is highly regulated?
- How can Medical Affairs enable its field team to engage more deeply with healthcare professionals (HCPs) on the go?
Moderna Medical Affairs sought out a software-as-a-service solution that would:
- Support a variety of workflows and research categorization using tagging and search filters.
- Host various libraries including research on COVID-19, RSV, Flu, among others– one that could scale alongside their growing pipeline.
- Offer custom fields allowing them to capture critical data points identified through regular literature reviews and updates.
Researching solutions
While researching potential solutions, the Medical Affairs team discussed their challenges with a colleague from Moderna’s Clinical team, who recommended ReadCube. The Clinical team had implemented the literature management platform a few years prior and found great success.
“Our colleagues strongly recommended ReadCube and explained that it is the gold standard solution,”
says Prashant Sable, Associate Director, Digital, Medical Affairs at Moderna.
The Medical Affairs team evaluated ReadCube’s capabilities and was immediately sold on its functionalities, value, and power to provide agile and trustworthy SCP integration. They discovered that ReadCube can also host all of Moderna’s searchable research reports and articles from multiple sources.
The ReadCube solution
The Medical Affairs team worked with ReadCube’s technical team to upload and organize their content into various libraries creating a now agile scientific communications platform.
Integrating third-party databases via ReadCube
PubMed is a third-party database containing over 37 million citations and abstracts of biomedical literature. ReadCube’s seamless integration with PubMed has been critical for Moderna’s Medical Affairs employees. By adding ReadCube’s browser extension, they’re able to seamlessly add content to the Medical Affairs library from PubMed and other research databases.
In addition to supporting PubMed and research databases, ReadCube allows the team to streamline their integrations with literature reviews, which in the past relied on third-party vendors, but now leverage generative AI. The team initially worked manually, with each vendor providing respective metadata along with critical data points identified throughout the review process. As the team has evolved to use custom GPTs for literature reviews and summaries, they are still finding their metadata seamlessly integrates into the respective ReadCube library via an imported CSV file.
For example, the COVID-19 library contains the standard metadata for each reference as well as additional critical data points (e.g., population, variants, and country). Thus, empowering the team to easily narrow and refine the content. User-friendly, collaborative, and searchable platform The Moderna Medical Affairs team found the overall ReadCube library creation and platform integration process intuitive, fast, and user-friendly—with limited training required.
User-friendly, collaborative, and searchable platform
The Moderna Medical Affairs team found the overall ReadCube library creation and platform integration process intuitive, fast, and user-friendly—with limited training required.
ReadCube’s search functionality makes it easy to find relevant literature, such as therapeutic areas, within libraries organized using tags or custom fields. Prashant Sable explains, “It’s like finding the right needle in a haystack.” ReadCube even allows users to filter out unwanted search results via the custom
search tool.
Sule and Sable used EndNote in the past but found ReadCube more robust and user-friendly. It helped them solve all of their team’s challenges and needs. For instance, Moderna Medical Affairs teams can easily collaborate and share content with anyone via the ReadCube platform.
“If your team is preparing a presentation, you can simply select what you need, even a whole category of research papers, then send it to a colleague’s inbox,” says Sable.
Moderna Medical Affairs now uses ReadCube as a tool for field enablement. While engaging and collaborating with a healthcare provider, Moderna Medical Science Liaisons can access figures, graphs, or research papers through an SCP content library integration with a simple mouse click or finger tap—all with citations in the spur of the moment.
Responsive account support and training
The Moderna Medical Affairs team has been pleasantly surprised by the level of customer service the ReadCube client engagement team continuously provides.
“ Michele and the team have always been really proactive, making sure that all our needs are met,” says Kennedy.
“ While it is true that ReadCube is easy to use, there are moments where you just want to phone a friend when you hit
a problem and customer support has always been incredibly responsive.”
The ReadCube team hosted multiple training sessions early on, which they recorded so new and existing Medical Affairs employees could learn how to use the platform. Employees can also refer to the content whenever they need a refresher. “It’s top-notch,” says Kennedy.
Results and future plans
While Moderna Medical Affairs is still evaluating ReadCube’s impact on the team’s performance, employee word-of-mouth feedback has been extremely positive.
“ People love the ease of use,” says Kennedy.
“ They say it’s a seamless user experience, and from our side, we’re completely satisfied.”
ReadCube continues to be a natural product fit to support the needs of Moderna’s Medical Affairs employees, and there have been many happy surprises along the way. In the future, Moderna plans to work with ReadCube customer support to expand new functionality as new product needs and platform features evolve.
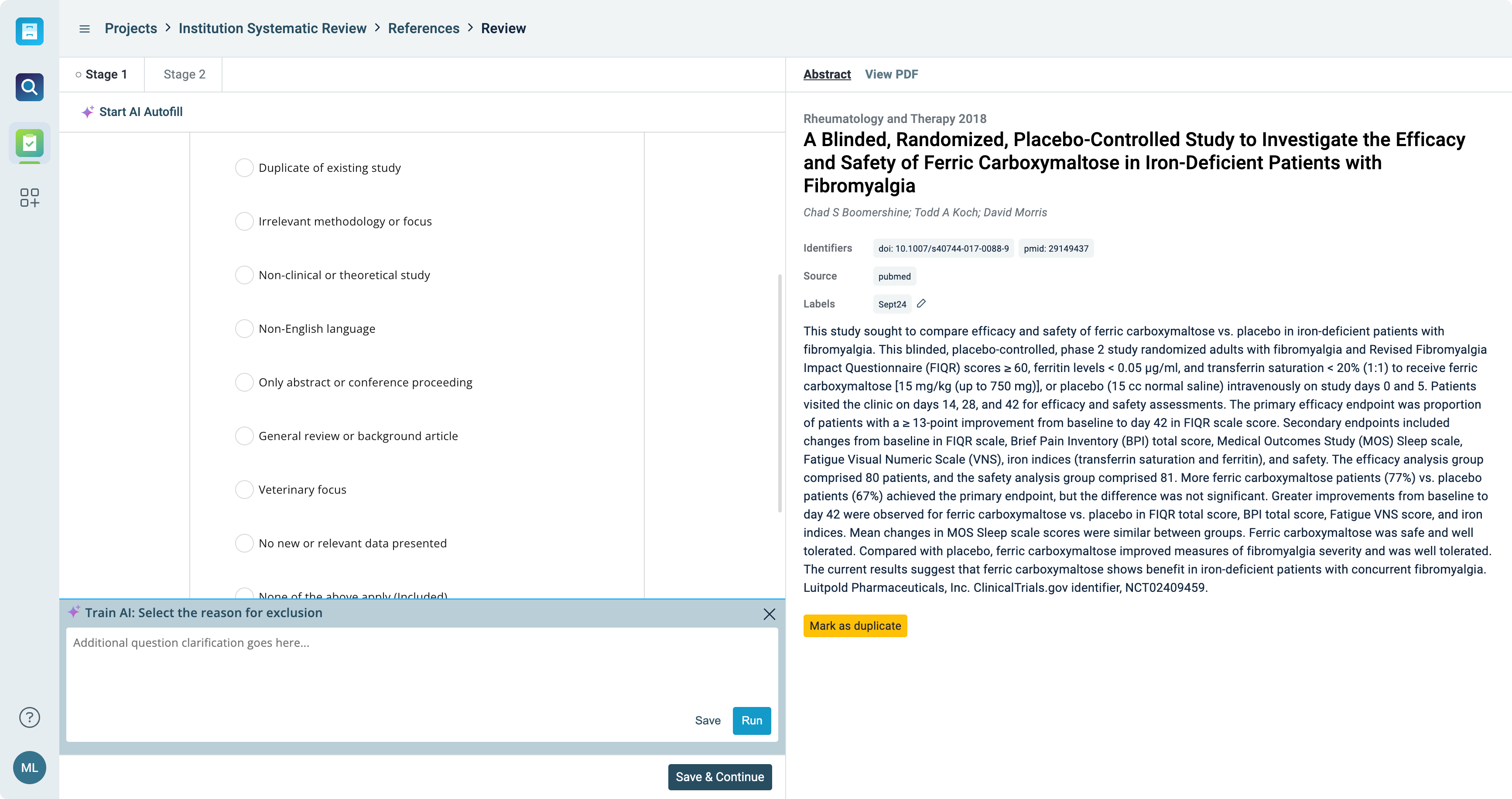
Build custom review workflows
- Implement multi-level screening for an in-depth literature review and screening
- Streamline the review process with your predefined inclusion and exclusion criteria
- Easily identify, manage, and review any conflicts in data entries when working with multiple reviewers, ensuring alignment and consistency
- Seamlessly integrate data from past reviews, ensuring continuity and time-saving efficiency
Depending on your department, industry, and goals, literature reviews vary greatly. ReadCube’s flexible and customizable Literature Review makes it simple for you to design the review that makes the most sense for your team.
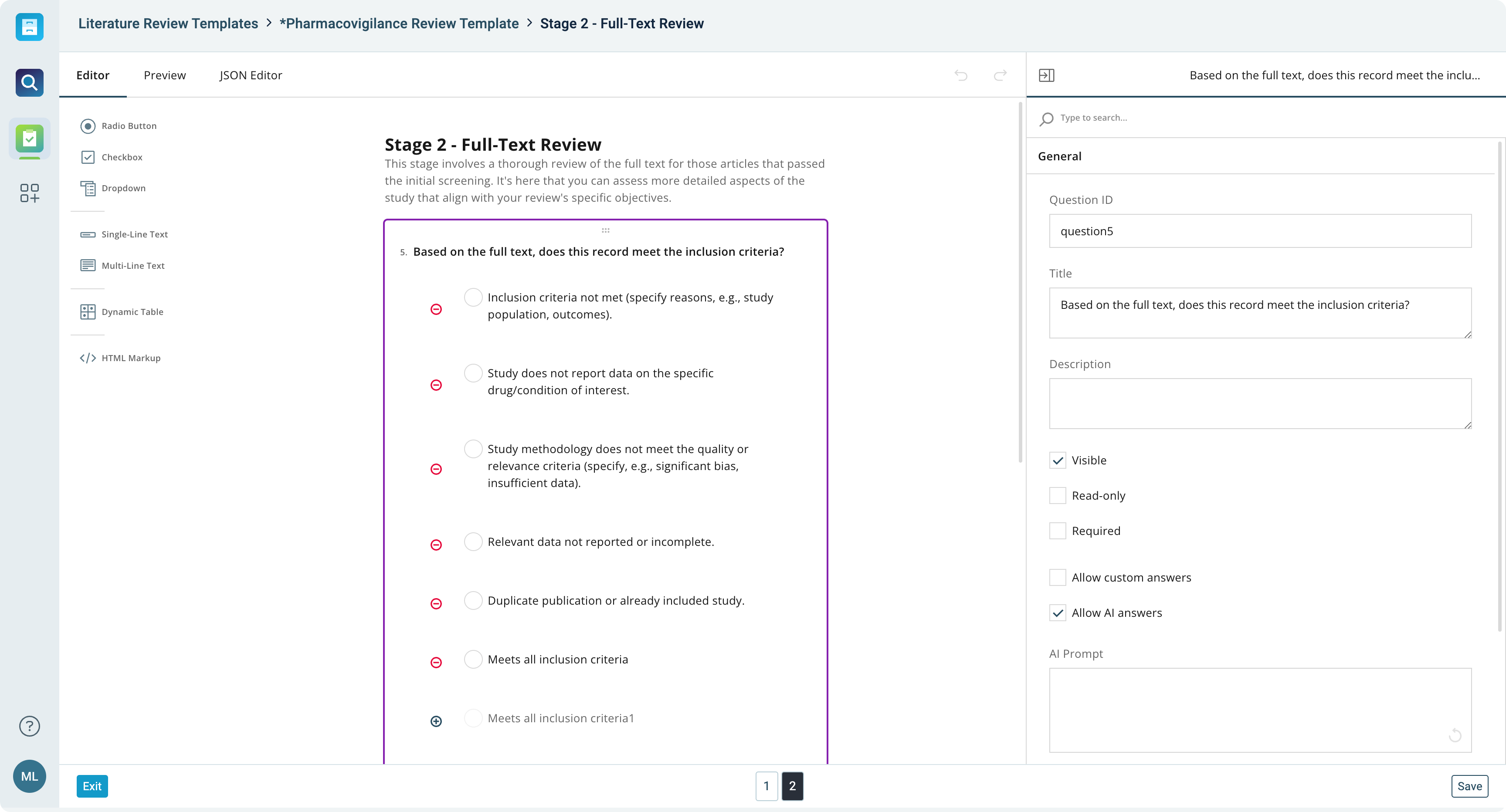
Ask the right questions
- Design and manage systematic review forms and questions with ease
- Add specific search results automatically to your review workflow
- Create subforms, select specific fields, implement conditional hidden fields, and manage shared fields
- Build advanced, calculated fields for enhanced accuracy in data assessment
Design the literature review workflow that actually works for your team with advanced and flexible form builder options.
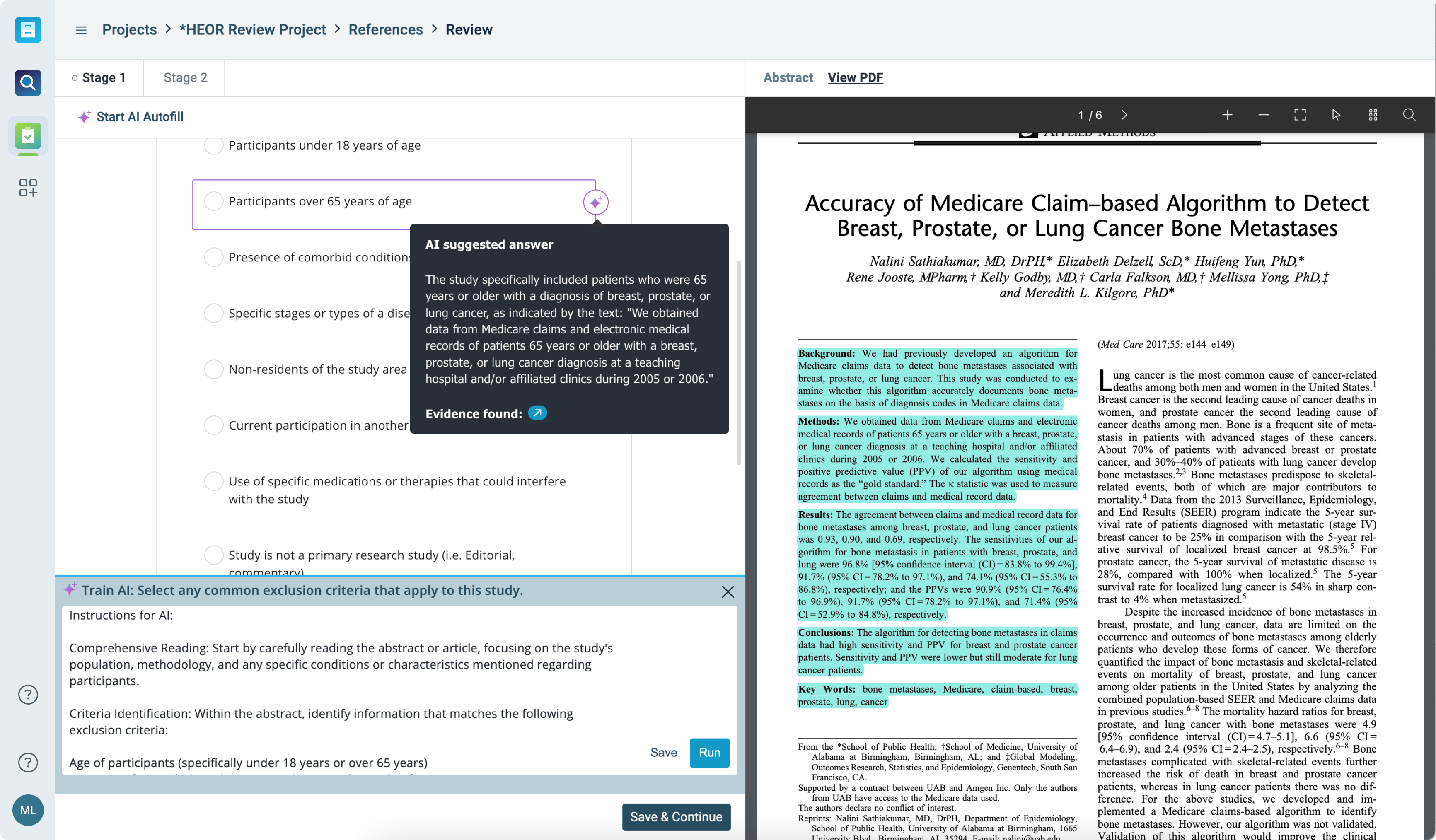
AI-enhanced review and analysis
- Make data-driven decisions faster with actionable AI insights that simplify complex reviews
- Automatically generate summaries, extract key data points, and create visual representations to support your analysis
- Leverage machine learning to identify patterns, gaps, and key insights across your literature set
Systematic reviews are faster, more organized, and enhanced by AI, allowing you to focus on generating insights, not managing paperwork.
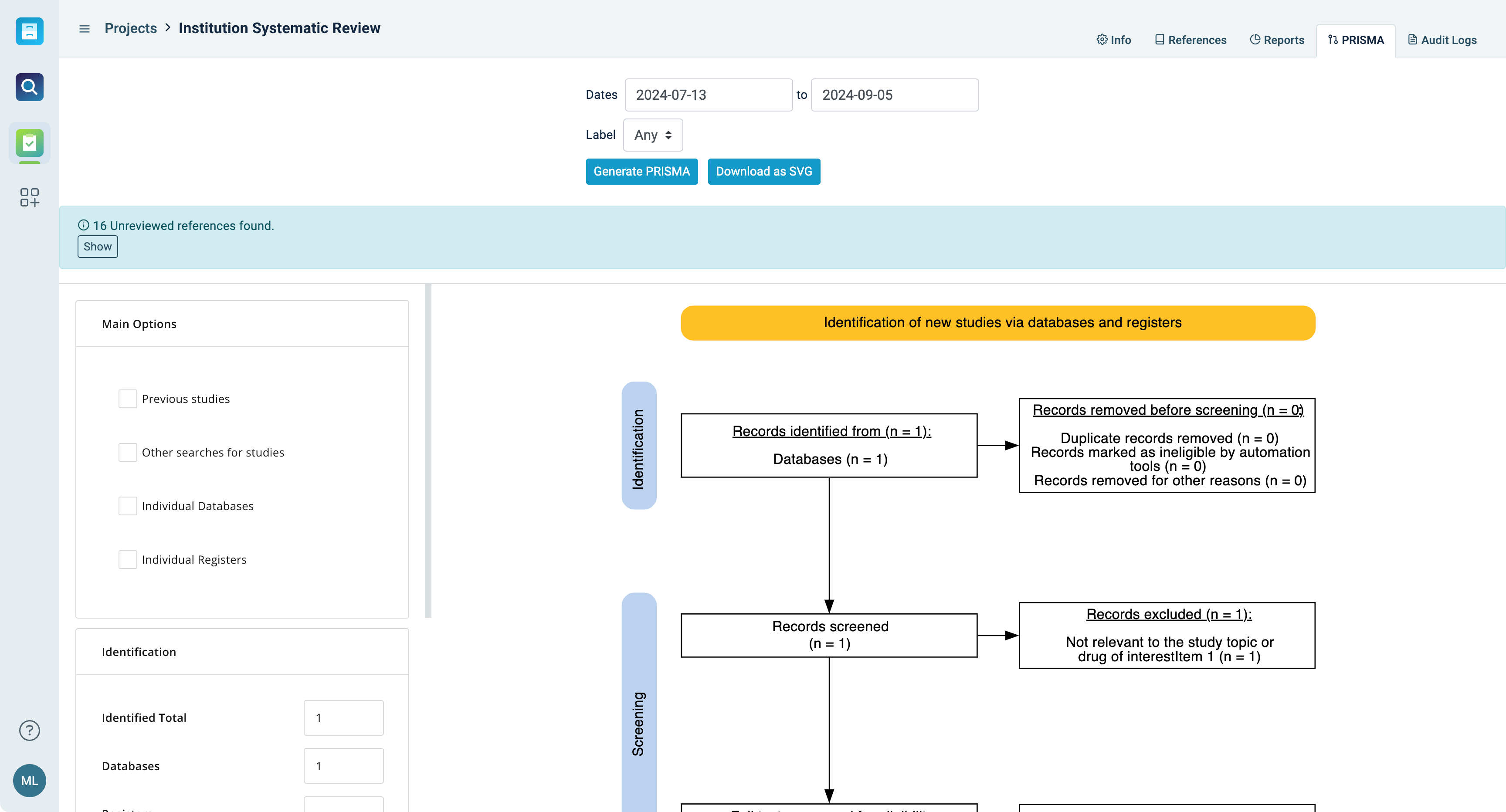
Share your findings
- Utilize PRISMA for structured reporting and visualization, providing detailed flow diagrams in editable formats
- Customize exports for reporting - including citation metadata, form data, and custom metadata fields
- Maintain a detailed audit log of all imports, changes, and identified conflicts, ensuring transparency and traceability
What’s the point of a review if it’s difficult to share results? Simplified and automatic reporting make it easy to share what you’ve learned - so your organization can make better evidence-based decisions.
Manage your reviews.
- Seamlessly import data using common file types such as RIS, CSV, and nBib from third party databases
- Automatically flag duplicates upon import or manually flag duplicates as part of your review
- Admin dashboards show an overarching view of all projects, with the ability to assign form templates, construct new form templates, delegate reviewers to specific projects, and manage the status of ongoing projects
- Reviewer dashboards show only the projects and citations relevant to a reviewer’s role. Understand current project status, pinpoint potential conflicts, and seamlessly access reporting features for exclusion, inclusion, and PRISMA
Ensure your team is on track and making progress with advanced governance features.